G.L. McLaughlin, J.M. Pascale, A. Amador, B. Barrett, P. Hahn, and S.F. Hensley &mdash The Australian Society for Microbiology, Inc. — Presented at the 1998 Annual Meeting and Exhibitions of The Australian Society for Microbiology, Inc.
Introduction
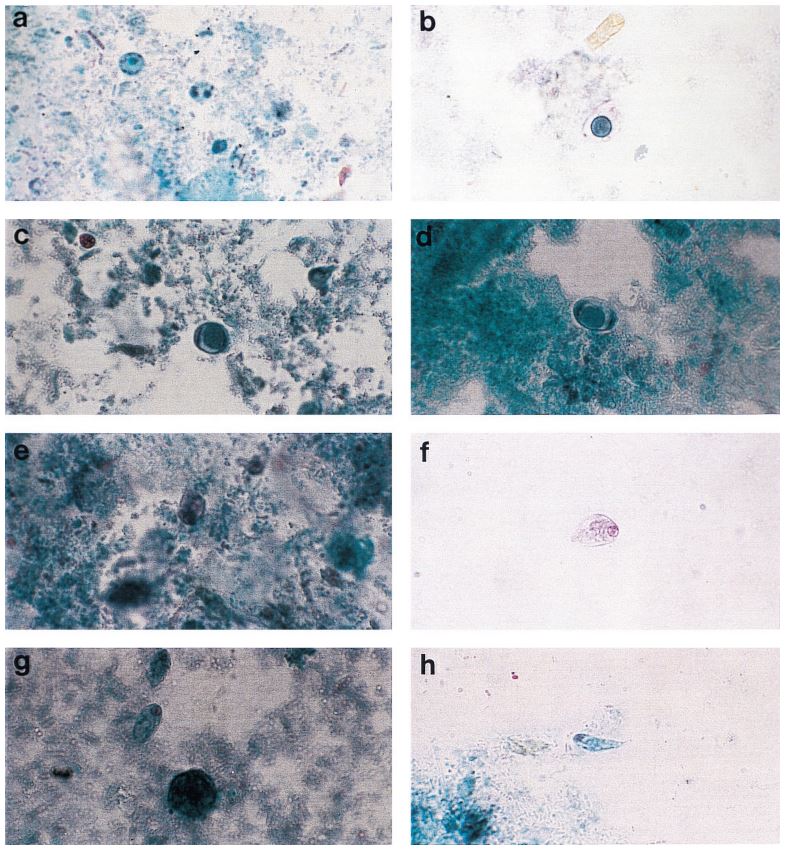
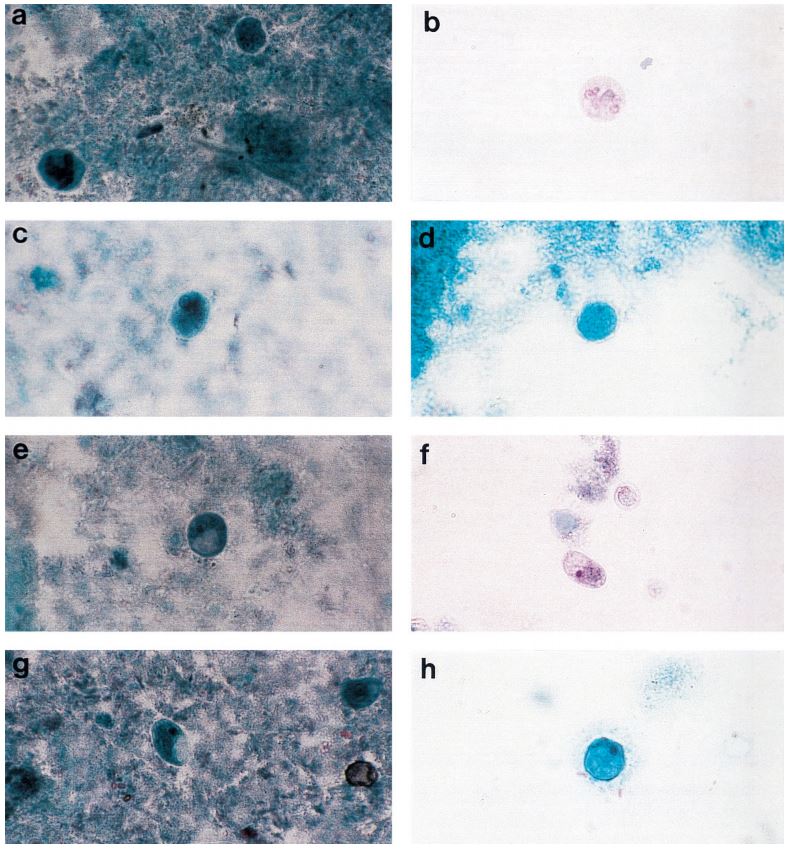
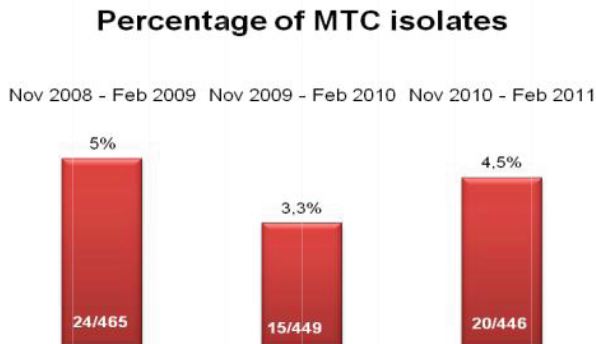
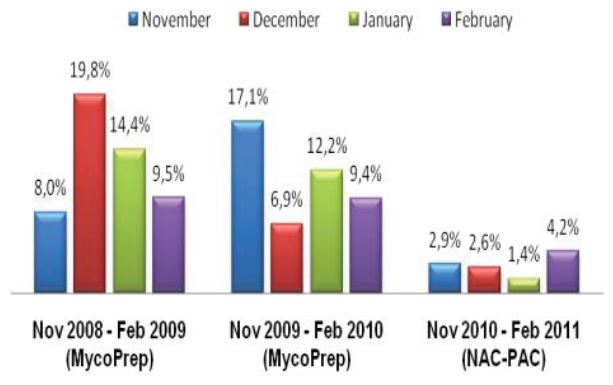
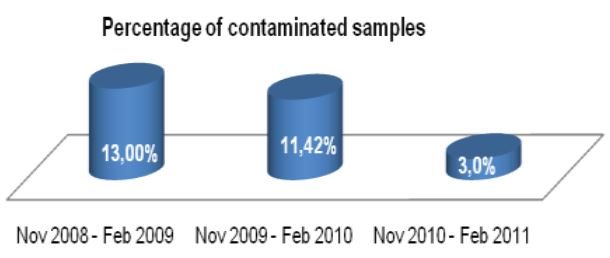
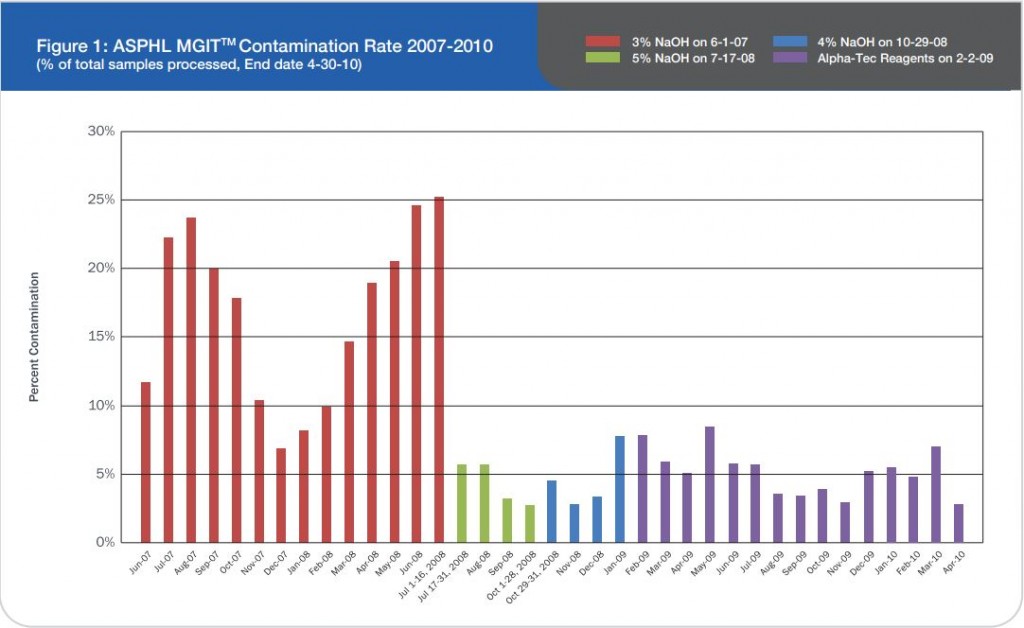
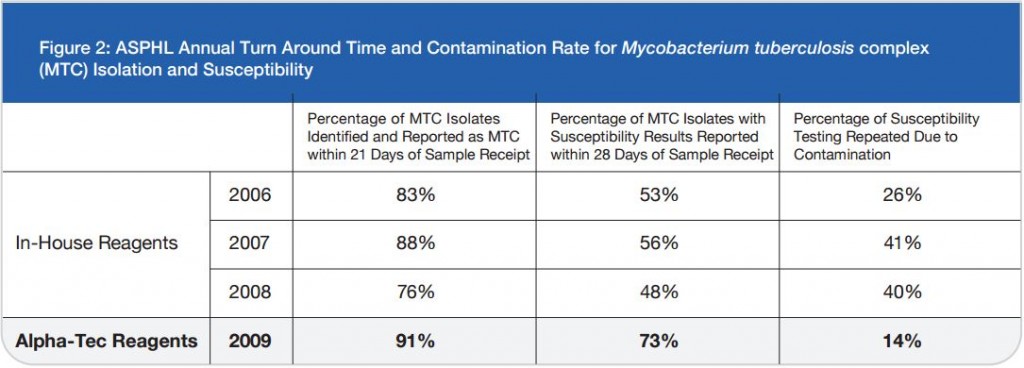
 The TechLab Giardia ELISA assay was evaluated with duplicate cultured Giardia spiked into feces and mixed with 8 different transport media and fixatives at 8000 cysts per 0.1 ml sample. The average O.D. readings of positive samples for both Enteric Set ETMTM and PROTO-FIXTM exceeded the average O.D. of all of the other fixatives tested.
The TechLab Giardia ELISA assay was evaluated with duplicate cultured Giardia spiked into feces and mixed with 8 different transport media and fixatives at 8000 cysts per 0.1 ml sample. The average O.D. readings of positive samples for both Enteric Set ETMTM and PROTO-FIXTM exceeded the average O.D. of all of the other fixatives tested.
Abstract
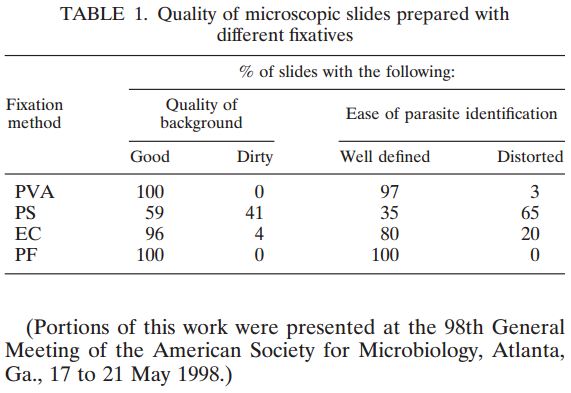
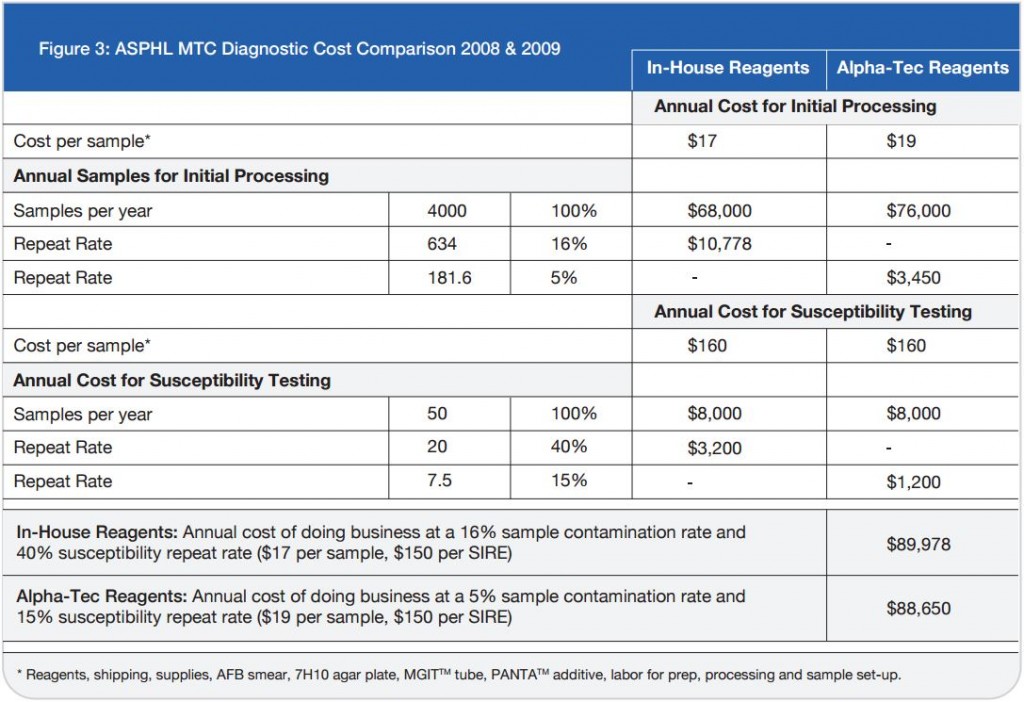
There is increasing interest in streamlining clinical parasitology laboratory procedures by employing ELISA assays and single-vial fixatives; it would be useful if such fixatives could be used in conjunction with ELISA assays. A new ELISA assay for Cryptosporidium from TechLab (Blacksburg, VA) was evaluated using a battery of fecal samples that had been examined microscopically in wet mounts and after acid fast staining, and by EIA assays, in parallel with commercial ELISA assays from Alexon and Remel. Sixty-five positive and 28 negative samples were correctly identified by both the TechLab and Alexon ELISA assays for Cryptosporidium, with no false positives or negatives, relative to scoring by EIA and/or light microscopy of acid fast and Trichrome stained slides. Remel’s ELISA’s showed high background readings using the chosen 60 minute initial incubation. The TechLab Giardia ELISA assay was also evaluated with duplicate cultured Giardia spiked into feces and mixed with Enteric Set ETMTM, PROTO-FIXTM, 10% formalin, SAF, MF, Parasafe®, EcoFix®, and LV-PVA, at 8,000 cysts per 0.1 ml sample. Average O.D. readings of positive samples decreased in the listed order of fixatives; negative controls included samples positive only for E. histolytica. In summary, the TechLab Cryptosporidium ELISA showed 100% sensitivity and specificity relative to Alexon’s ELISA and microscopic diagnosis, and both ELISA assays were compatible with stool preserved in several one-vial fixatives.
Parasafe® is a trademark of Scientific Device Laboratory, Inc., Des Plaines, IL, USA.
EcoFix® is a trademark of Meridian Bioscience, Inc., Cincinnati, OH, USA.
ETMTM and PROTO-FIXTM are trademarks of Alpha-Tec Systems, Inc., Vancouver, WA, USA.
©2015, Alpha-Tec Systems, Inc. All rights reserved.
CC14-354, Effective Date: 16 Jan 2015, M-PO-AUS.A