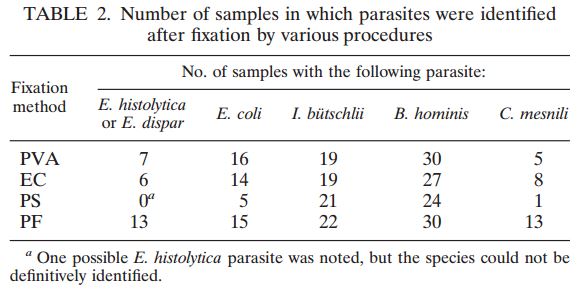
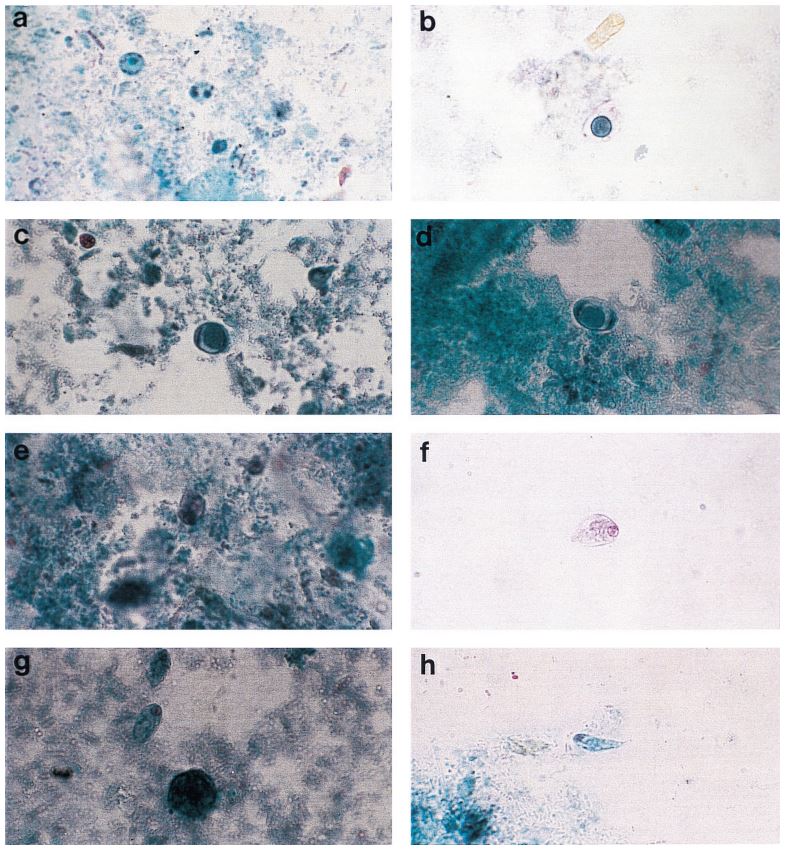
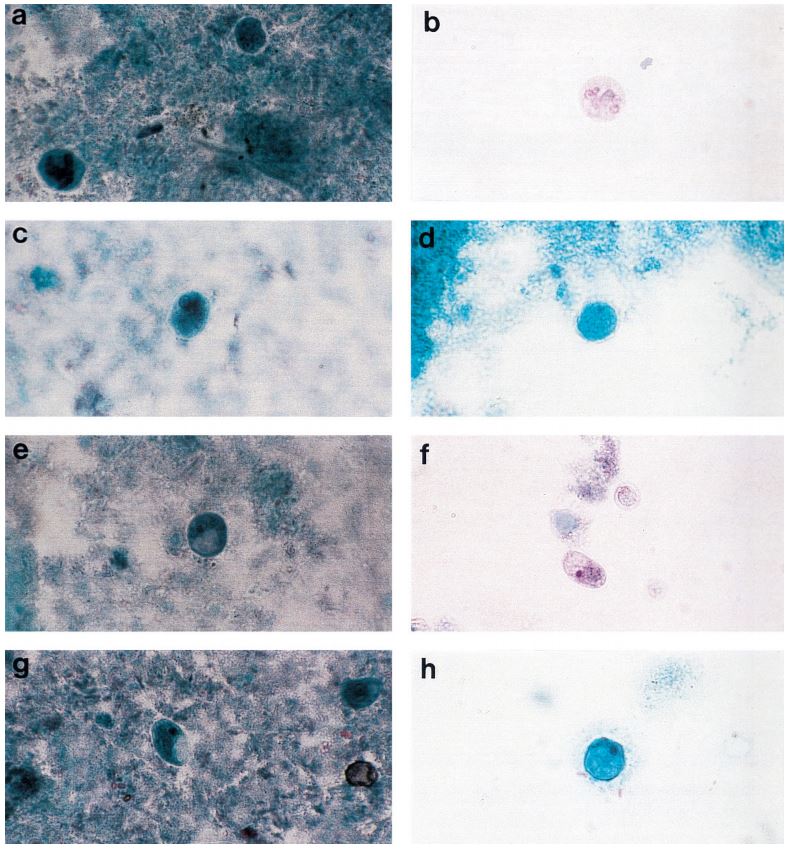
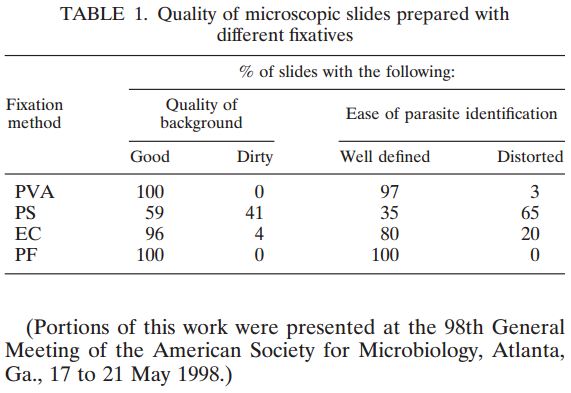
Cammaggi Anna, Molinari Gian L., Mantovani Monia, Brunelli Maria G., Fortina Giacomo (Department of Microbiology and Virology, “Maggiore della Carità” University Hospital, Italy) — Presented at the 2011 European Society for Mycobacteriology
Cammaggi Anna, Molinari Gian L., Mantovani Monia, Brunelli Maria G., Fortina Giacomo (Department of Microbiology and Virology, “Maggiore della Carità” University Hospital, Italy) — Presented at the 2011 European Society for Mycobacteriology
Introduction
The majority of biological samples intended for mycobacteria detection are usually contaminated with oral flora resident bacteria. The mycobacteria growth, if compared to the other bacteria is considerably slower; therefore, a decontamination procedure is required of the sample before streaking on suitable culture media. Decontamination is intended to reduce the microbial contamination at the lowest possible levels so as not to interfere with the growth of mycobacteria. The decontamination procedure may affect the viability of the mycobacteria, so this should not be too aggressive in the method and timing.
Materials and Methods
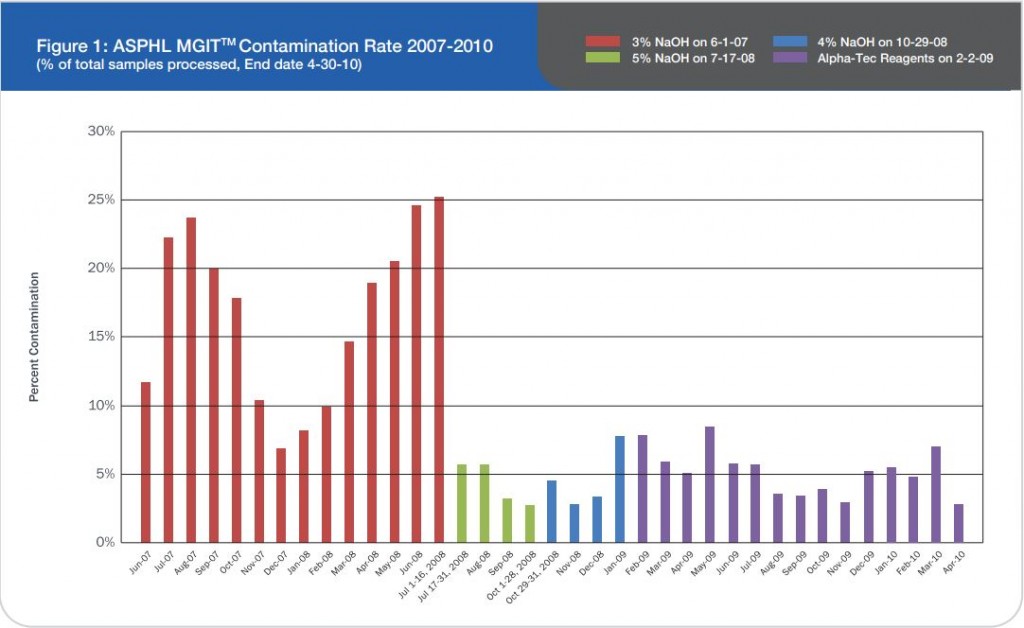
Between November 2010 and February 2011 our laboratory processed 446 biological samples using the NAC-PACTM RED (Alpha-Tec, USA) (Fig. 1). We compared the percentage of contaminated samples by using NAC-PAC RED and the percentage of contaminated samples obtained in the previous years (2008-2009) using MycoPrepTM
(Becton, Dickinson and Company – BD). We evaluated only the results obtained using the liquid culture medium MGITTM 960 (BD) rather than the solid media, as the non-radiometric liquid media has a very rich composition and therefore is the most contaminated of the liquid media. Both methods use N-acetyl-L-cysteine (NALC) together
with sodium hydroxide (NaOH) and sodium citrate as the digestant / decontaminating reagent. NAC-PAC RED also contains a pH indicator. A pH indicator is incorporated into the digestion and decontamination reagent to monitor the pH throughout the decontamination and buffering procedure, allowing the laboratory technologist to visually see when neutralization has been achieved. pH neutralization is indicated by a color change from red/pink to colorless.
Results
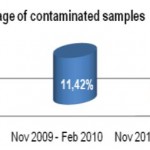
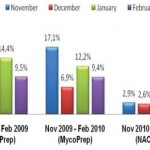
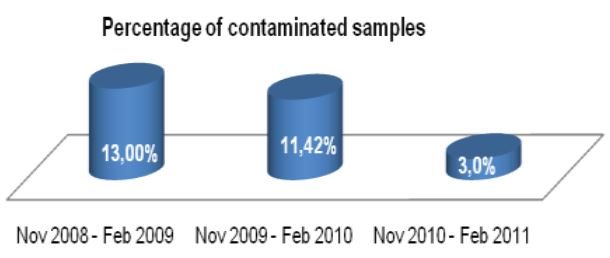
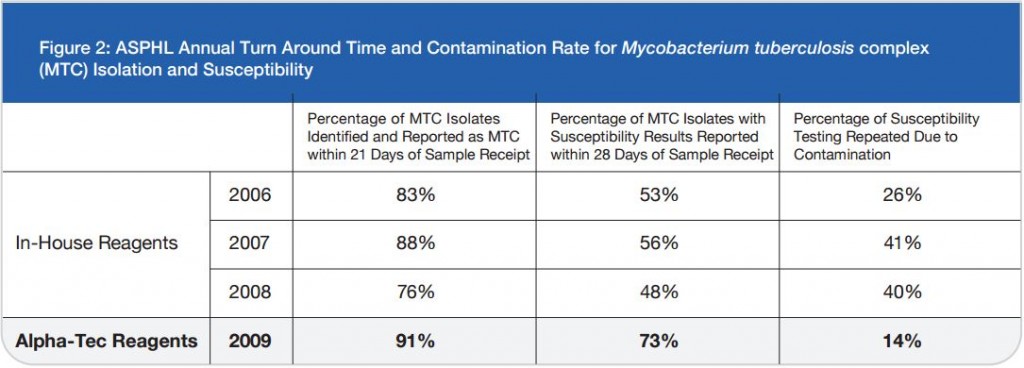
On 446 analyzed samples (Nov. 2010 – Feb. 2011), only 13 (3.00%) were contaminated. Over the same period in
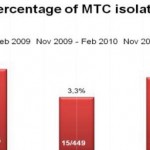
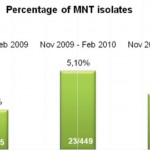
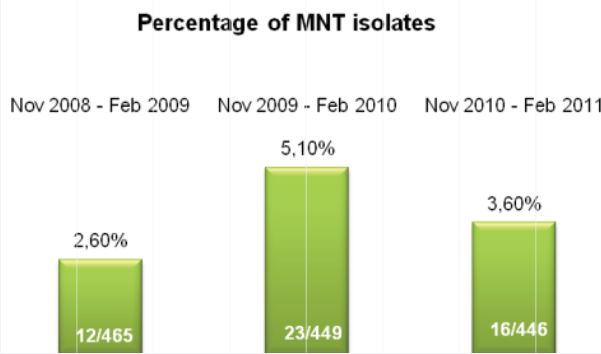
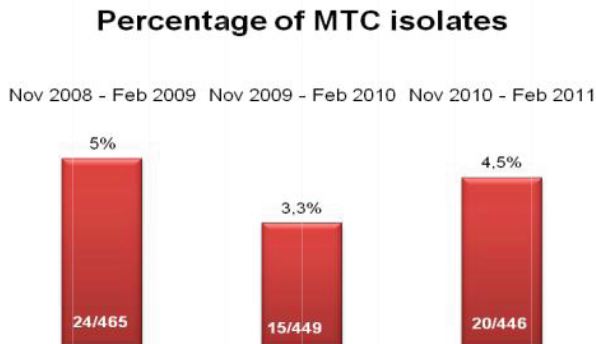
2009, on 449 analyzed samples, 49 (11.42%) were contaminated. In 2008 we analyzed 465 samples and 61 (13.00%) were contaminated (Fig. 2, Fig. 3). We also considered the number of positive samples for Mycobacteria in the same periods in order to detect if the decontamination methods had an impact on the viability of Mycobacteria. The results of both methods were comparable; in fact, in the period between November – February, the positive MTB samples were 24 (5.0%) in 2008/2009, 15 (3.3%) in 2009/2010, 20 (4.5%) in 2010/2011 (Fig. 4). Positive samples for NTM were 12 (2.6%) in 2008/2009, 23 (5.1%) in 2009/2010, 16 (3.6%) in
2010/2011 (Fig. 5).
Conclusion
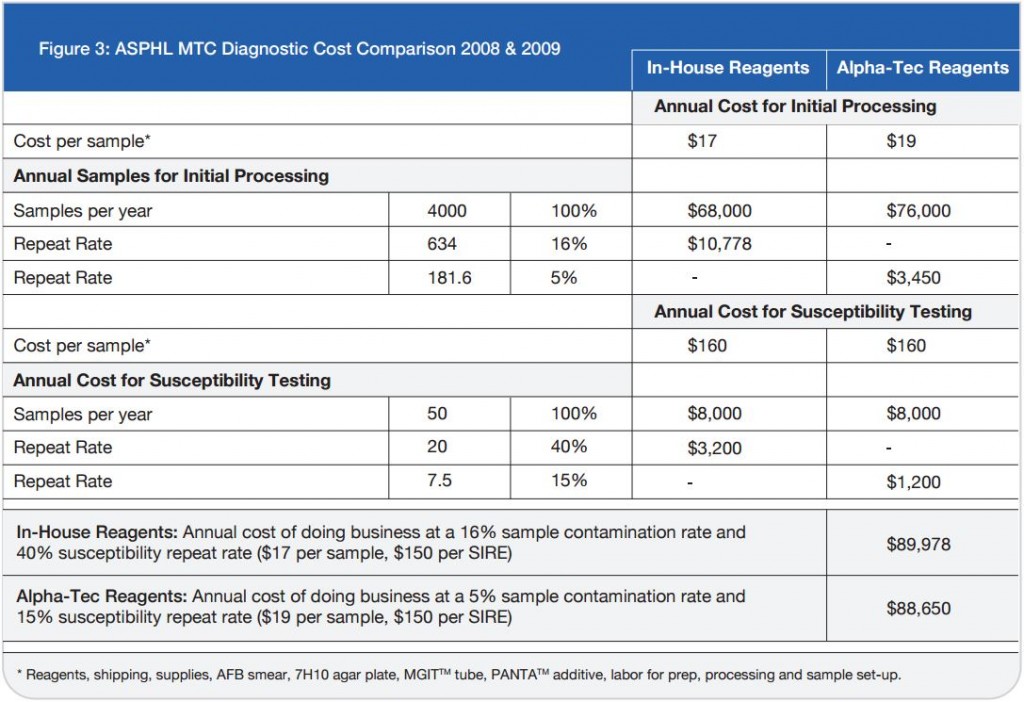
In our Country, the centralization of diagnostic procedures is evolving, even for microbiology diagnostic laboratories and this has a direct effect on the contamination rates. NAC-PACRED, in this study, has demonstrated to be a valid means of overcoming this problem, by reducing the contamination rates from 13% to 3%. This reduction of contamination has led to a significant reduction of sample re-processing. These improvements directly affect the quality of the care and treatment received by the patient. This reduction moreover has afforded our labouratory a reduction in the labour commitment and cost
NAC-PAC™ is a trademark of Alpha-Tec Systems, Inc., Vancouver, WA, USA. ©2014, Alpha-Tec Systems, Inc. All rights reserved.
CC14-239, Effective Date: 28 Aug. 2014, M-PO-ITA.A