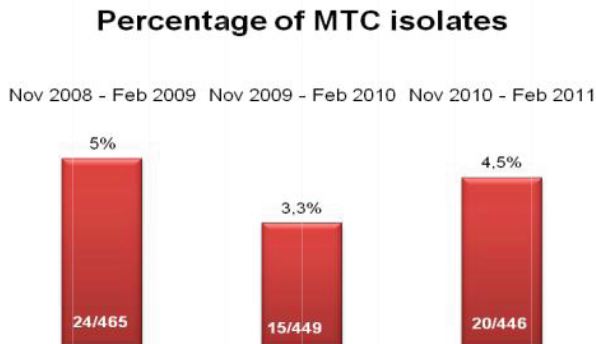
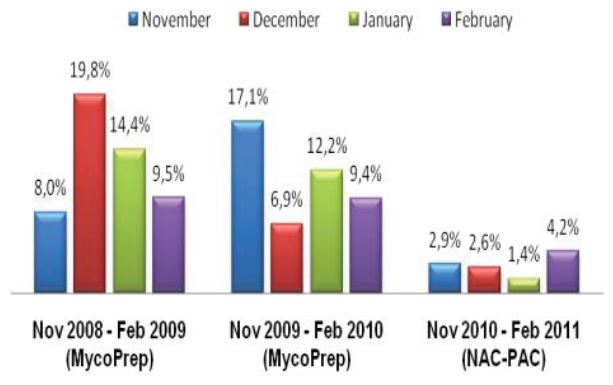
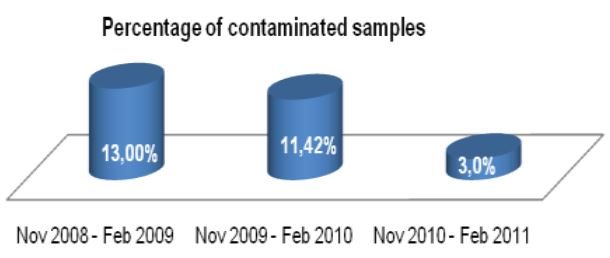
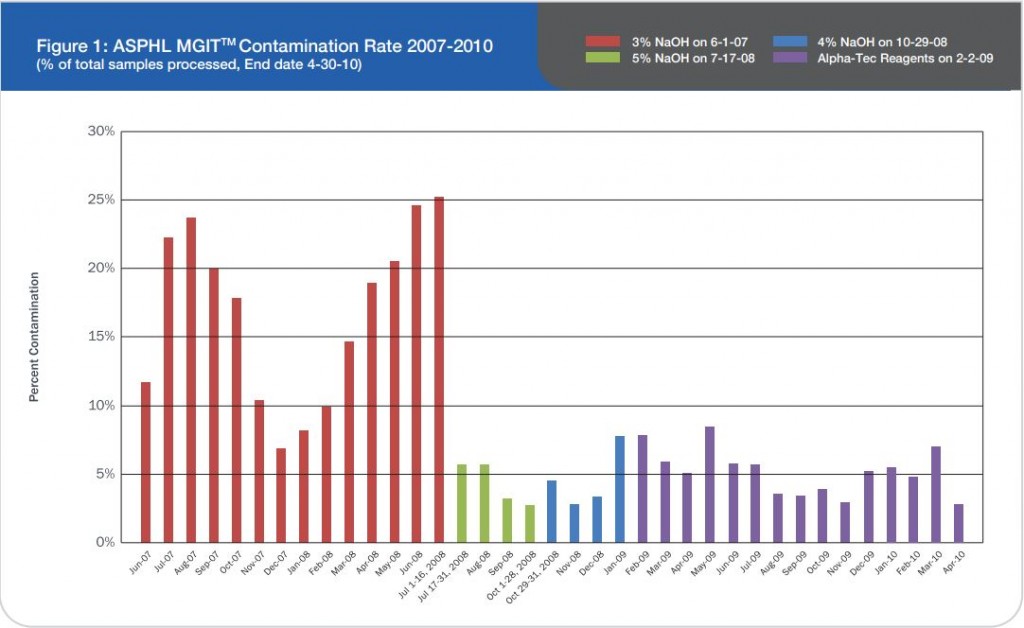
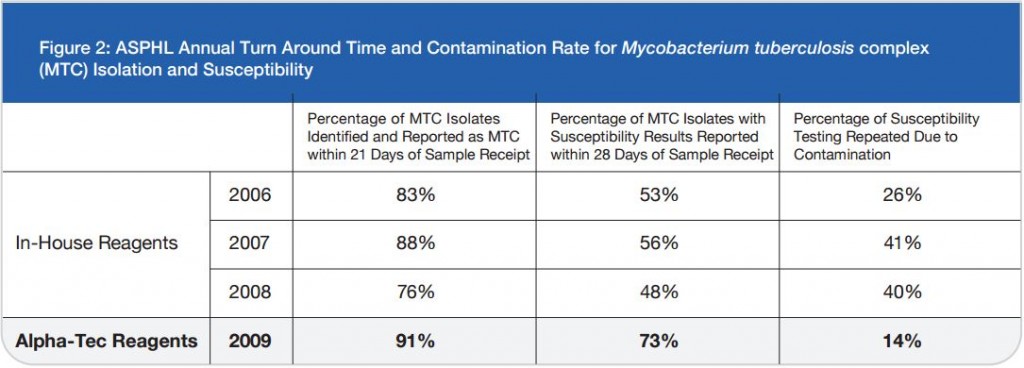
During microscopic diagnosis for certain diseases, laboratory technicians handle quality control slides containing potentially hazardous organisms such as tuberculosis on a daily basis. Generally, they are careful to avoid exposure to these organisms; however, many may be unaware of potential risks associated with handling these slides. Lab professionals utilizing established products, such as quality control slides containing Mycobacterium tuberculosis, risk TB exposure that could potentially lead to contracting the disease – a disease that is subtly, but consistently, on the rise.
Tuberculosis has been, and continues to be, the leading cause of death in many third-world countries such as India, Africa, and the Philippines. While the number of TB cases in the past 23 years has been on the decline in the United States, recent reports indicate a reversal in this trend due in part to an increase in immigration from countries that have a high incidence of active TB. These rising numbers should, in turn, prompt re-evaluation of existing TB testing methods with the goal of improving overall accuracy and safety for healthcare workers.
Methods used for TB diagnosis are decided primarily by economic considerations. One of the least expensive methods, and the one used most commonly worldwide, is Slide Smear Microscopy (SSM). SSM begins with collecting a sample of the patient’s sputum, smearing it on a slide, heat-fixing it, and then staining it along with a quality control slide containing mycobacteria. After that, the lab tech can place both slides under a microscope for investigation. Use of the quality control slide creates a double-checking system, clarifying that the staining procedure was performed properly and thereby ensuring the most accurate diagnosis possible. This procedure corresponds to policies and safety protocols that are always in place and, ideally, should be strictly followed by technicians and enforced by laboratory managers.
Yet, even though SSM is the process of record worldwide for diagnosing TB, its overall safety within labs should be questioned, especially for lab professionals in third-world countries where little in terms of safety protocol is taught and even less is enforced. Here in the US this is also a fundamentally important question since AFB quality control slides (Acid-fast bacilli) which contain M. tuberculosis are commonly used and passed from technician to technician, often without the use of gloves or any other protective gear. Sometimes these slides are even distributed to the general public at trade shows where many non-laboratory workers, such as marketing and sales professionals, are in attendance to learn more about products and services within the medical and pharmaceutical industries.
Fighting this rise in active TB cases relies on accurate diagnoses of the disease, which in turn requires laboratory staff to handle patient samples and diagnostic devices potentially containing TB. If not handled properly, risk of contracting the disease is possible. Interestingly, existing documentation pertaining to TB infection via the handling of these slides is difficult to find; not much has been written about lab-related safety issues regarding the use of TB slides since the 1990s. In fact, few surveys of laboratory-acquired infections have been undertaken at all. Most reports are of small outbreaks in specific laboratories. However, there was one instrumental 1957 study that established international safety codes of practice for those working with TB. It stated that British medical laboratory workers had a risk of two to nine times that of the general population of acquiring tuberculosis. Furthermore, other studies have shown that tuberculosis is the most common lab-acquired infection out there.
An added consideration when exploring this topic is how TB is spread. While the majority of tuberculosis cases worldwide occur through airborne transmission, studies within the past 60 years have proven that cutaneous transmission of tuberculosis, referred to as CTB (from skin-to-skin contact) is possible, and documented cases exist. Cutaneous tuberculosis is a specific strain of M. tuberculosis whose pathology is unique, and treatment for it differs from airborne strains of the disease. Subsequently, it is treated as more of a dermatological issue and is often misdiagnosed.
A definitive answer regarding safety levels associated with handling slides inoculated with M. tuberculosis in labs remains to be found. Within the limited studies available regarding lab-acquired TB, the consensus is that slides using M. tuberculosis are infectious and should be viewed as potentially dangerous for those handling them. Companies developing diagnostic products need to evolve from standard, run-of-the-mill producers of these diagnostic tools to active, aggressive risk management intercessors, leading the way in integration of cutting-edge research and helping laboratories adjust to new tools and procedures.
One company located in Washington State, Alpha-Tec Systems, has reduced the risk of lab-acquired TB by using Mycobacterium scrofulacium on their quality control slides instead. M. scrofulaceum has been proven to be just as effective as using M. tuberculosis on slides for validating the stains used for diagnosing active TB. According to Anne Frontella, Business Development Manager at Alpha-Tec, M. scrofulaceum slides are an equivalent and much safer alternative. Through the development of these innovative slides known as QC1™ AFB Slides, Alpha-Tec is paving the way within the diagnostic community.
However, meeting the growing demand for safer, increasingly accurate diagnostic testing involves on-going, cumulative research. Updating education and safety guidelines is also necessary, along with enforcement of existing policies. In tandem with such progress, manufacturers of diagnostic products have a responsibility to actively pursue solutions that may reduce such risks. Hopefully, these changes will improve AFB processing, thereby decreasing the possibility of the spread of this highly communicable disease.
MK-WEB-RESOURCES-STUDIES-MYCO-TB-001.A CC18-104, Effective Date: 30 March 2018