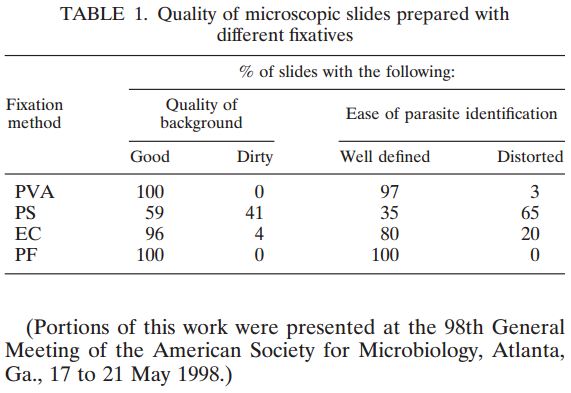
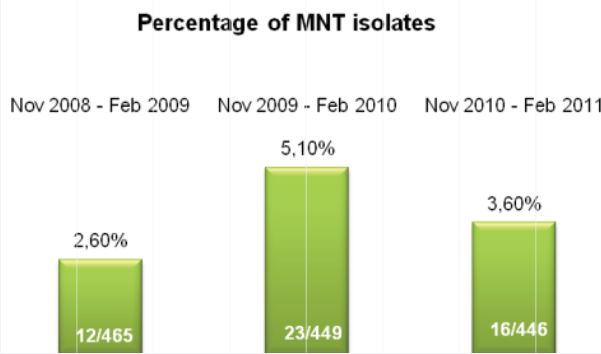
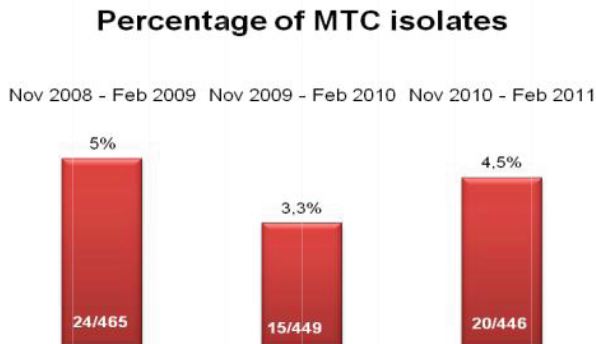
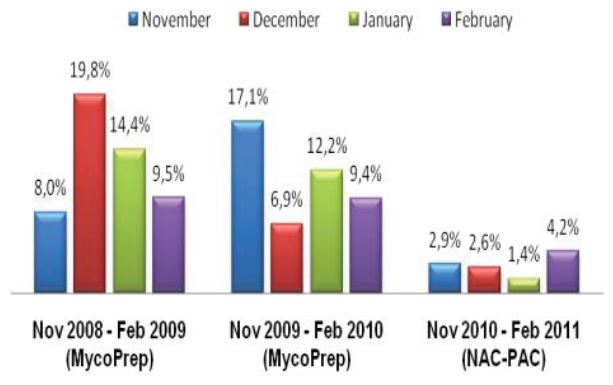
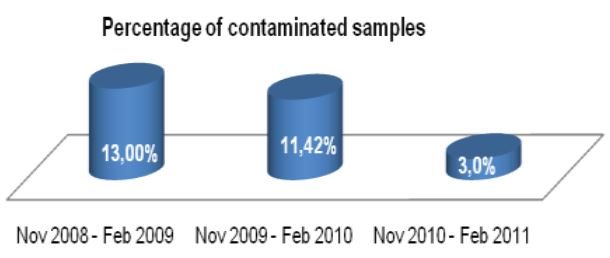
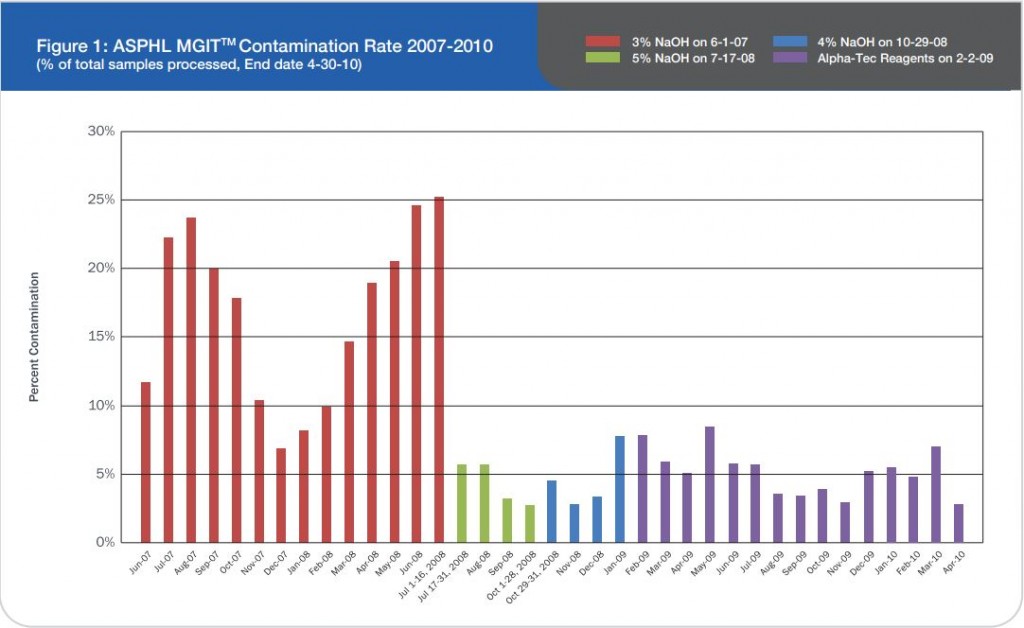
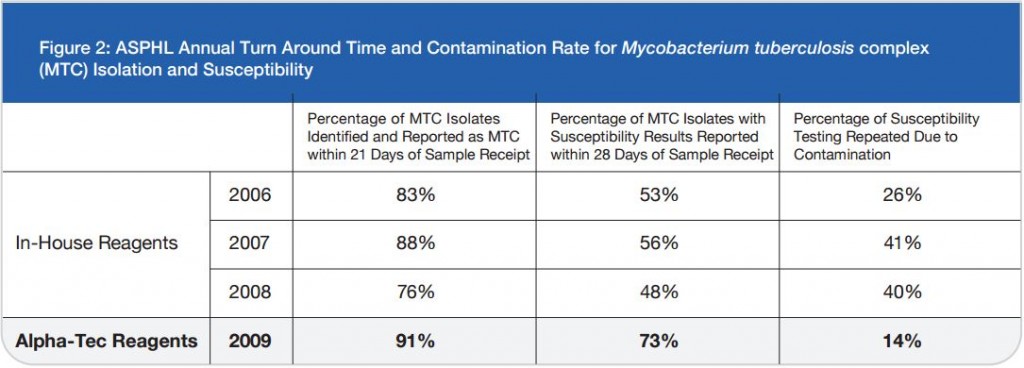
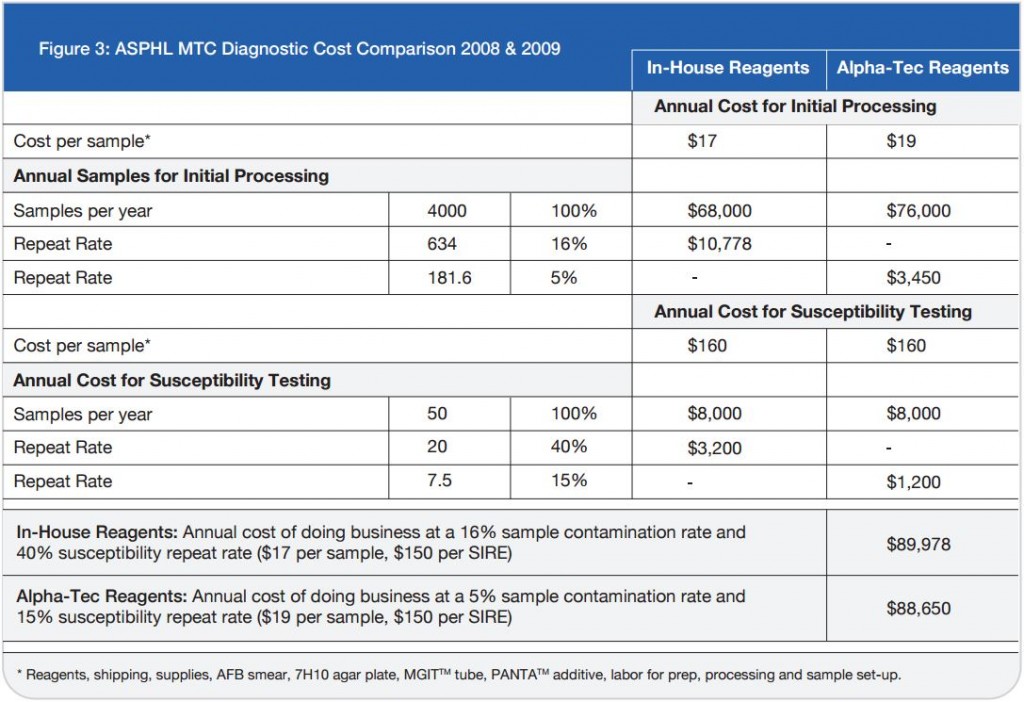
Microbix Quality Assessment Products (QAPs) are manufactured from native pathogens so that they closely mimic the patient samples. These liquid ready-to-use aliquots or sample swabs are inactivated and non-hazardous for immediate use to control elution (swabs), extraction, amplification, and detection steps of NATs. For over 30 years, Microbix has been a critical manufacturer of antigens for OEM tests and viral material for proficiency testing and external quality assurance for clinical laboratories.
REDx Controls are quality control products intended to evaluate laboratory testing performance, procedures, and daily workflow of detection devices, while PROCEEDx Controls are RUO IQ/OQ/PQ control products that are used to ensure that the device complies with its specifications during setup and for teaching and training laboratory personnel. Depending on the organism, these controls are used with nucleic acid and/or antigen assays that detect infectious disease pathogens in patient samples. Critical for their use in Clinical Quality Management System (QMS) workflows, REDx and PROCEEDx Controls improve the diagnostic quality outcome by early detection of deviations from desired assay performance. Independent third-party control materials such as Microbix REDx and PROCEEDx Controls are highly recommended for use in addition to/instead of control materials supplied by the reagent or instrument manufacturers to ensure the validity of diagnostic results.